A key trait of most ceramic coatings are their hydrophobic properties. As a formulator and manufacturer of ceramic coatings and cleaners, hydrophobicity is an important factor to us. And if it’s important for you too, come join us as we enter the fourth installment of “The Science of” by the IGL R&D team.
Read previous entries by IGL Coatings R&D team here. Topics include an in-depth explanation of VOC, Graphene, and Ceramic Coating.
In this installment, we will be discussing the theory behind the hydrophobic behavior of a material and how it’s measured. Hydrophobic is a term derived from the Latin words “hydro-” (water) and “-phobia” (fear), which is used to describe a surface with low affinity to water or repel water. Hydrophilic comes from “hydro-” (water) and “-phile” (love), which indicates that the surface has a high affinity to water or is easily wet by water. The shape of the water droplet on a surface change depending on the degree of affinity of the coating surface to the water, as shown in Figure 1.
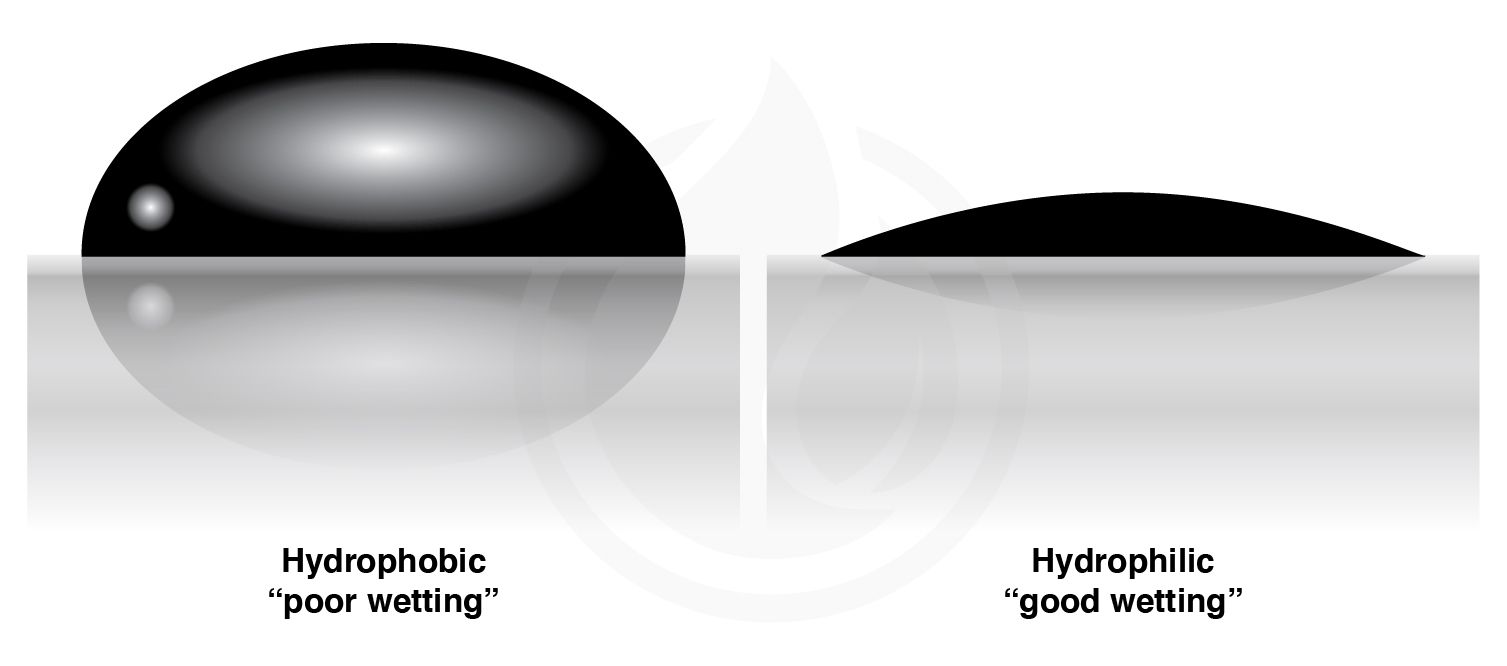
Water is used as an indicator to measure how hydrophobic and/or hydrophilic a coated surface is. As such, it’s important to understand the nature of water.
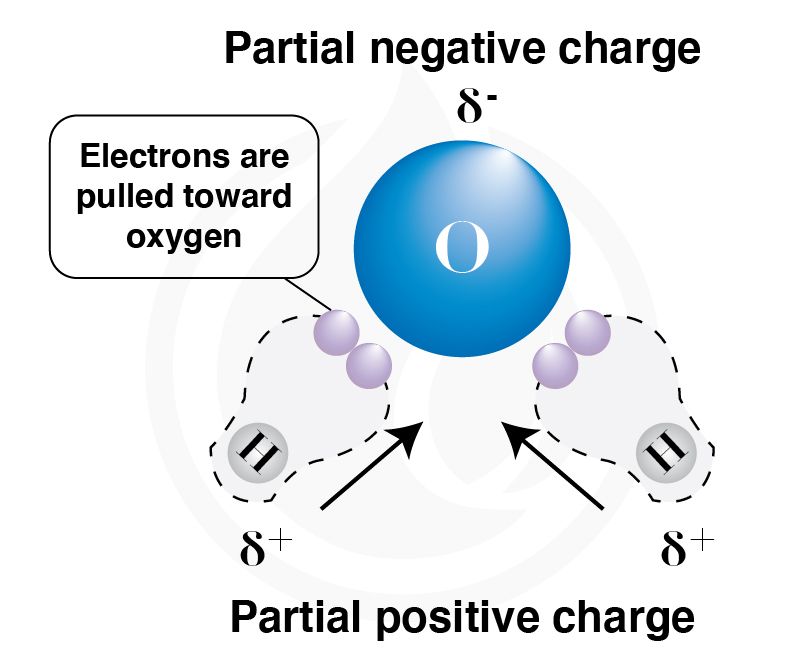
Water is a polar molecule, which means it carries a partial charge between atoms. In a water molecule (H2O), the oxygen molecule is more electronegative, which allows it to attract the electron pairs in each bond towards itself. This creates a partial negative charge on oxygen and a partial positive charge on the hydrogen atom, as shown in Figure 2.
Water also has a relatively high surface tension at around 72 mN/m. Surface tension is defined as the tendency of fluid surfaces to shrink into a minimum surface area. A real-life example can be observed when rain is collected on lotus leaves as displayed in Figure 3, commonly referred to as the lotus effect. Water adheres weakly on the leaf surface and strongly to itself (due to high surface tension) creating water clusters into beads, forming the smallest possible surface area to volume ratio.

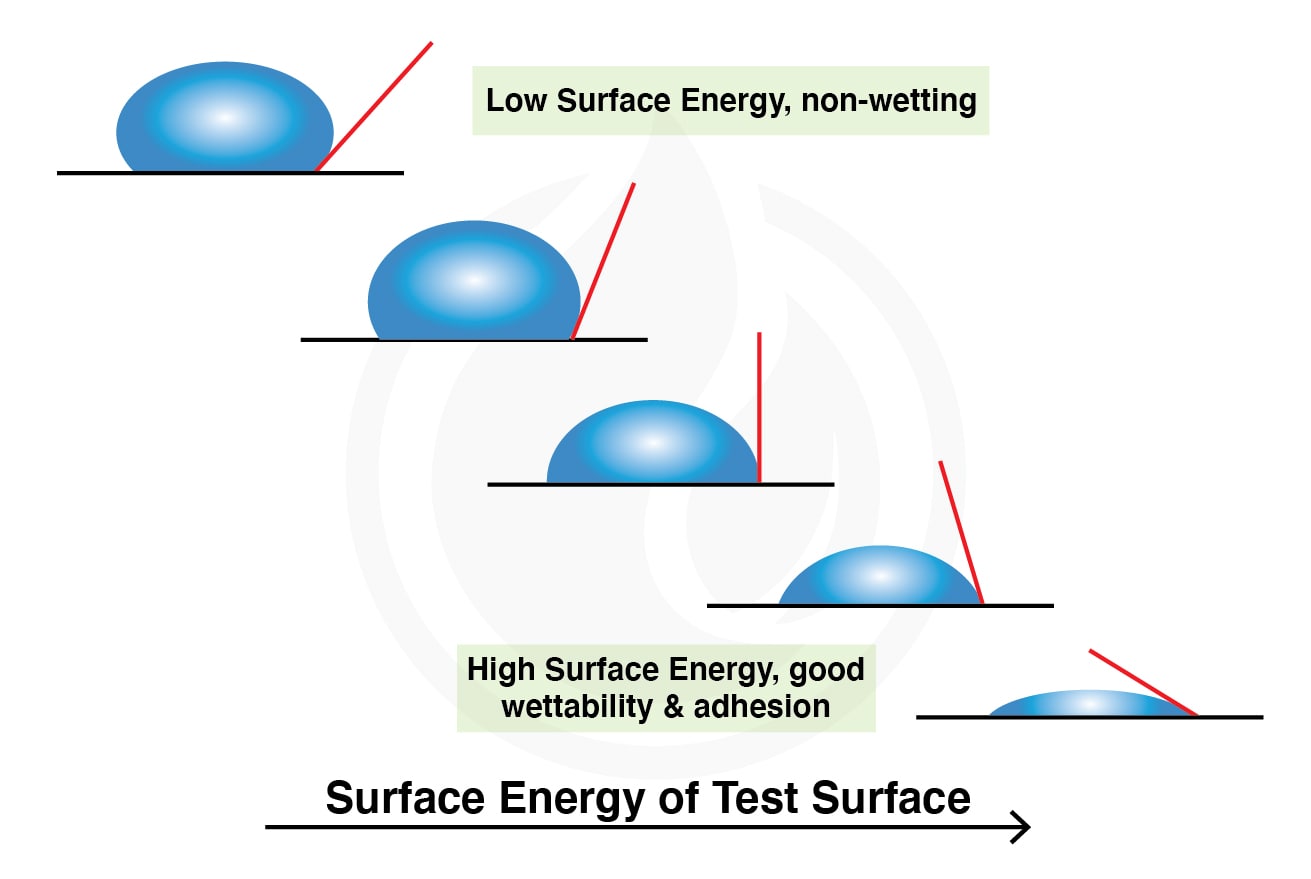
Hydrophobicity and hydrophilicity are co-related with surface free energy (SFE) which will affect the wettability of the surfaces. SFE is defined as a quantitative measurement of intermolecular force at the surface which is independent of the liquid used. SFE can also be considered as the surface tension of a solid. By understanding the SFE of the solid, we are able to predict the behavior of liquid on the surface.
Referring to the example of the lotus effect in Figure 3, water droplets adhere weakly on the surface of the lotus leaf. This phenomenon also can be explained by both SFE and surface tension of liquid applied, as this involved the interaction between solid (known as surface) and liquid applied. When the SFE of the solid/surface is low, it is difficult to be wetted by water (poor wetting). The same theory applies to our ecocoat series. When the ceramic coating is coated on the paintwork of the automotive, it creates a surface with low SFE. The ecocoat surface thus is able to repel water and dirt to protect the paintwork, known as hydrophobic. On the other hand, bare metal and glass surfaces possess high SFE which showing good wetting, thus showing hydrophilic originally.
As an example, the ecocoat window plays its role to lower the SFE of the windshield/windscreen, as the rain is unable to wet the windshield. This provides better visibility and clarity to the driver. Figure 4 displays the relation between SFE and surface wettability.
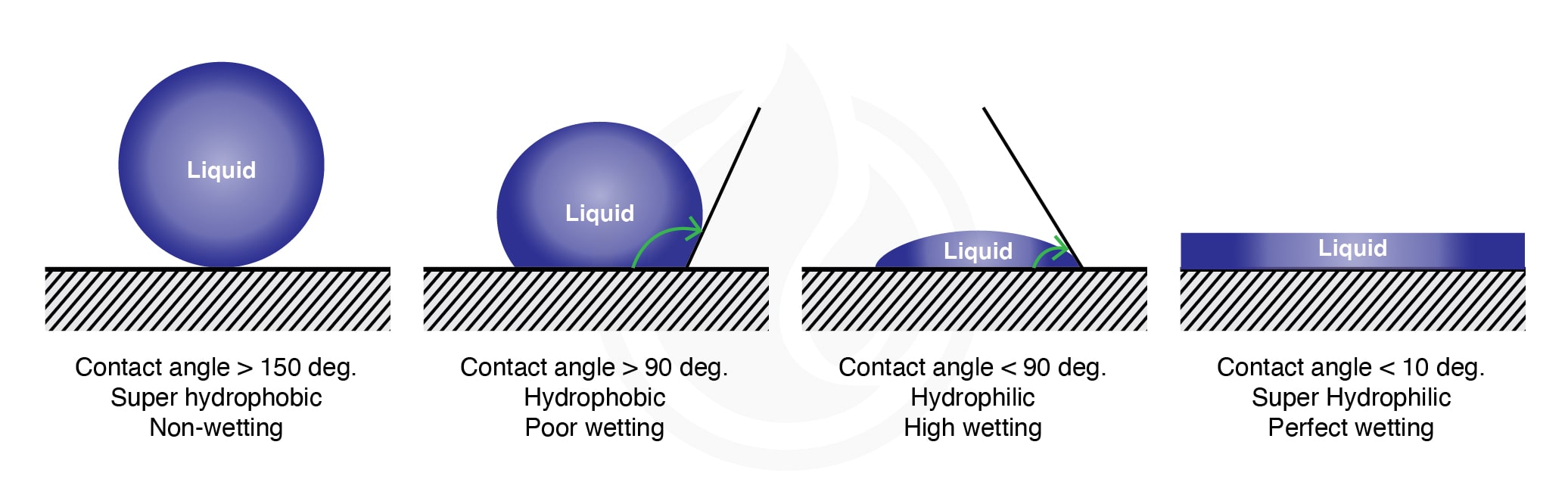
How are hydrophobicity and hydrophilicity measured?
Both hydrophobicity and hydrophilicity can be measured by using water contact angle measurement (WCA). In short, when the WCA is below 90°, the surface is defined as hydrophilic. When the WCA is above 90°, the surface is defined as hydrophobic. Meanwhile, if the WCA is >150° and <10° or almost zero, we defined them as super-hydrophobic and super-hydrophilic, respectively.
IGL Coatings’ newly upgraded Water Contact Angle Meter assists our R&D team during the development of both super-hydrophobic and hydrophilic coating by measuring the WCA of the prototype, illustrated in Figure 6.
This specialized equipment enables our R&D team to verify the improvement of WCA between uncoated surfaces and surfaces coated with our prototypes and plays an important role in the quality control of the current products. Every batch of the ceramic coating produced undergoes a series of quality checks before proceeding to be packed. This ensures good quality and functionality of the products from us to our consumers.
The measurement of the WCA is one of the most crucial in-house testing carried out on our ecocoat series.

To learn more about the eco-friendly coatings produced, formulated, and manufactured by IGL Coatings, visit www.iglcoatings.com for more information. You can also email us at marketing@iglcoatings.com for further requests and articles.


